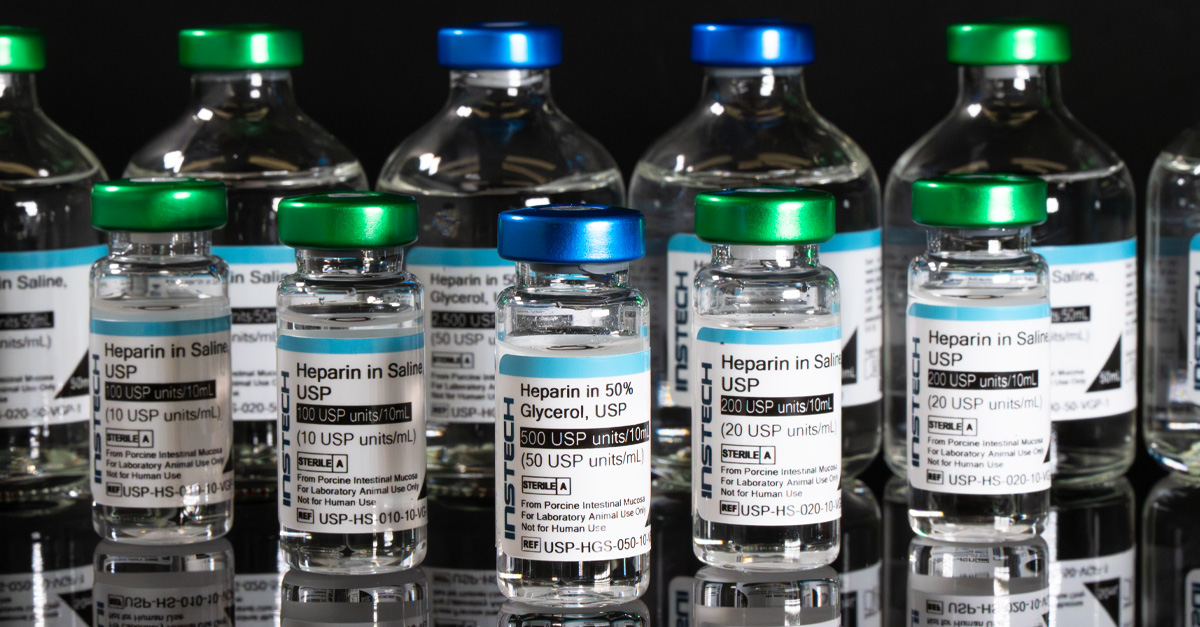
USP - the gold standard
 The gold standard for compound quality is “pharmaceutical-grade”: meeting the standards set by the US Pharmacopeia (USP) or other regional pharmacopeias for chemical purity. Look for "USP" in the product name on the label; it is not enough that the solution was made from pharmacteutical-grade components - it needs to have been prepared according to USP standards.
The gold standard for compound quality is “pharmaceutical-grade”: meeting the standards set by the US Pharmacopeia (USP) or other regional pharmacopeias for chemical purity. Look for "USP" in the product name on the label; it is not enough that the solution was made from pharmacteutical-grade components - it needs to have been prepared according to USP standards.
If you are injecting novel pharmaceutical compounds, why would you risk your research by flushing them in with solutions of unknown quality? Heparin in particular has a notoriously “twisted” supply chain, starting with pig farms in China. Contaminated heparin harmed hundreds of patients in 2008, leading the FDA to require improved testing to receive the USP designation. And yet non-pharmaceutical-grade heparin remains readily available from Fisher Scientific and other suppliers.
AAALAC International states that using pharmaceutical-grade compounds is preferred in some situations and required in others, if available. If you are using non-pharmaceutical-grade flush or lock solutions, your IACUC should require a justification.
NIH's Office of Animal Welfare states: OLAW and USDA agree that pharmaceutical-grade substances, when available, must be used to avoid toxicity or side effects that may threaten the health and welfare of vertebrate animals and / or interfere with the interpretation of research results.
See their Guidelines for Use of Non-Pharmaceutical Grade Compounds in Laboratory Animals for additional details.
Sterility is critical
 In addition to the chemical purity, you must be certain that your flush and lock solutions are sterile. Non-sterile solutions can cause patency problems at a minimum, or worse infections which could compromise your research and the health of your animals. Sterile USP solutions will have been prepared according to the USP <797> standard for compounding sterile pharmaceutical preparations. If you are preparing solutions in your lab starting with sterile components, you must follow careful procedures to make sure they remain sterile until they are injected. For example, syringes with tips that are not sealed will not maintain sterility.
In addition to the chemical purity, you must be certain that your flush and lock solutions are sterile. Non-sterile solutions can cause patency problems at a minimum, or worse infections which could compromise your research and the health of your animals. Sterile USP solutions will have been prepared according to the USP <797> standard for compounding sterile pharmaceutical preparations. If you are preparing solutions in your lab starting with sterile components, you must follow careful procedures to make sure they remain sterile until they are injected. For example, syringes with tips that are not sealed will not maintain sterility.
Interested in learning more about lock and flush solutions?
Read the Guide to Catheter Lock and Flush Solutions for Laboratory Animal Research