 On the surface an IV self-administration (IVSA) study is just like any other drug infusion study: you need to deliver a dose from a syringe pump, through a swivel and tether system to an indwelling catheter, typically the jugular vein. However, the nature of these studies, for addiction research or abuse liability, place unique requirements on the equipment. In fact, IVSA has helped push development to the benefit of other types of studies that use the same equipment, such as glucose clamping, infusion toxicology and blood sampling. Key considerations:
On the surface an IV self-administration (IVSA) study is just like any other drug infusion study: you need to deliver a dose from a syringe pump, through a swivel and tether system to an indwelling catheter, typically the jugular vein. However, the nature of these studies, for addiction research or abuse liability, place unique requirements on the equipment. In fact, IVSA has helped push development to the benefit of other types of studies that use the same equipment, such as glucose clamping, infusion toxicology and blood sampling. Key considerations:
- First, long-term patency is critical. Studies can last several months, so if you are losing catheter patency before your study is complete you will have to add extra animals, intervene surgically or possibly even re-run your study.
- Second, you are moving animals in and out of the operant chamber frequently, so the system must be simple to connect and disconnect. Doing so must not place undue stress on the animal or allow the catheter flow path to be open to air or bacteria.
- Third, these are sensitive behavioral studies so handling stress caused by the equipment will impact the data. Often you would like the animals to be socially housed when they are not in the chamber, so the equipment must allow for that.
If your IVSA equipment is failing along any of these measures—if you are losing patency, if it takes you more than a few seconds to connect or disconnect the tether, or if your animals are stressed or restrained—it is time to consider a change.
Disclaimer: Instech manufactures the Dacron mesh buttons, polysulfone buttons, Vascular Access Harnesses™ and Vascular Access Buttons™ referenced in this article.
Contents
IV Self-Administration Equipment for Rats
IV Self-Administration Equipment for Mice
IV Self-Administration Equipment for Rats

Chambers
Systems from Med Associates (such as part no MED-008-CT-B1) are nearly universal, though occasionally Panlab, Coulbourn, Lafayette Instrument, TSE or homemade systems are seen. The chamber should always have a responsive counter-balance (MCLA) to take up slack in the tether. This will allow for unrestrained movement and prevent bites and tangles, which are more of a risk with self-administration studies because the animals are often hyperactive.
Tethers
Tethers, or "leashes" as they are uniquely referred to in the IVSA community, include the line from the pump, a swivel to allow the animal to turn, the line to the animal, and a connection to an implanted catheter, are critical to the success of the overall system. There have been significant advances in these tethering systems over the past several years.
 Med Associates PHM-131 |
Back-mounted cannula connector pedestal (CamCaths IVSA28; Med Associates PHM-131; PlasticsOne / P1 Technologies / Protech 313-00BM, SAI SBD-06-30). This device has been used for IVSA for many years. A stiff mesh is implanted under the skin and the pedestal is suitable for long term use. Connection can be time consuming (tubing must be connected manually to an open tube and then spring threaded over to protect it) and it is not possible to use aseptic technique. However, the pedestal is much better than older systems where externalized catheters had to be connected with couplers. Group housing is possible with a screw-on metal cap. CamCaths in Cambridgeshire UK has ceased operations so availability may be limited. |
 Instech DC95BS |
Dacron mesh skin button (Instech DC95BS, Med Associates PHM-123D, SAI SBD-02). Instech developed this product nearly 30 years ago for long-term implantation. Tissue will grow into the mesh over time, but it was designed for permanent connection to a tether so connection and disconnection is awkward and opens the catheter line to contamination and air. Therefore, it is not a good product for IVSA. |
 Instech VAHR1H/22 |
Harness. Instech originally developed the rat harness (CIH95) in 1995 working with Covance Laboratories as an improvement on cloth jackets. It allowed a rat with a simple externalized catheter to be connected to a swivel and tether system after surgery. In 2006, we added a small external port to simplify the connection and in doing so created the first closed tethering system, the Vascular Access Harness™ (VAHR1H/22 (formerly VAH95AB)). Harlan Laboratories (now Inotiv) reported that this device significantly improved patency, allowing them to reliably conduct three month infusion studies. The ease of connection made the VAH™ useful for IVSA studies, but with several disadvantages: a segment of the catheter is externalized which can lead to evaporation and loss of patency when fluid is static, the bands around the animal need to be checked frequently, and there is no ability to group house animals. |
 Instech VABR1B/22 |
Instech Vascular Access Button™ (VAB™) (Instech VABR1B/22; distributed Med Associates as PHM-VABR1B/22). In 2009, we worked with Janssen Pharmaceuticals in Belgium to combine the benefits of the CamCaths button with the port in the VAH™ to extend patency and simplify connections in their IVSA studies (Teuns et al, 2014). The result, the Vascular Access Button™, solved many of the problems with previous systems and has been successful not only in IVSA but in many other applications such as blood sampling for pharmacokinetics, glucose clamping and infusion toxicology. We updated the design in 2016, creating models with two, three and four ports and adding magnets to further simplify connection. The VAB™ is a closed system—the catheter is subcutaneous rather than exteriorized which allows for long-term patency and is an animal welfare refinement. Group housing is made possible with a magnetic protective cap. |

Rat Catheters

The catheter that is placed in the vessel is also critical.
-
 First, it should have a round tip to help with insertion and decrease trauma to the inner lining of the vessel walls. (You should never trim a round catheter tip and never use a beveled tip catheter.)
First, it should have a round tip to help with insertion and decrease trauma to the inner lining of the vessel walls. (You should never trim a round catheter tip and never use a beveled tip catheter.) -
It should have suture collars or beads at the appropriate spot to keep it in place long-term and guide you to the ideal insertion length. For jugular vein catheterization the catheter tip should be just above the right atrium of the heart. Instech has catheters with insertion lengths matched to body weights and a surgical SOP to ensure ideal positioning.
-
The catheter should be made of polyurethane—PE is too stiff for vessels, silicone is too slippery to make reliable connection with the VAB™ connector.
-
It should have an inner diameter (ID) that is ideal for the device to which it is connecting (.61-.63mm to connect to 22ga tubing). Instech uses a multiple-step process to ensure our catheters meet standards for consistent ID which alleviates issues with inadvertent disconnection from the VAB™.
-
And without fail it should be sterilized; ethylene oxide gas exposure in Tyvek® pouches is really the only way to sterilize polyurethane catheters and any implanted component such as the VAB™.
Rat Catheter Connection
 The connection between the catheter and the Vascular Access Button™ connector deserves special attention with IVSA studies because you are trying to extend patency as long as possible. With the proper inner diameter-to-outer diameter fit it should be nearly impossible to pull the tubing off the connector; the PU tubing will shrink down as it stretches. (That said, be sure to leave enough slack in the catheter because with your improved system your animals may be growing larger than they have in the past.) However, if you force fluids through the catheter, either manually during a flush or with the syringe pump during a dose, and that catheter is blocked or kinked, you can generate very high pressures. If you push hard enough the weakest point in the line will leak. Sometimes the culprit is an external connection, but it is physically possible to cause a leak, and even a disconnection, where the catheter connects to the button. The best solution to this is to make sure these forces don’t occur, but if warranted you can take extra steps to secure this connection:
The connection between the catheter and the Vascular Access Button™ connector deserves special attention with IVSA studies because you are trying to extend patency as long as possible. With the proper inner diameter-to-outer diameter fit it should be nearly impossible to pull the tubing off the connector; the PU tubing will shrink down as it stretches. (That said, be sure to leave enough slack in the catheter because with your improved system your animals may be growing larger than they have in the past.) However, if you force fluids through the catheter, either manually during a flush or with the syringe pump during a dose, and that catheter is blocked or kinked, you can generate very high pressures. If you push hard enough the weakest point in the line will leak. Sometimes the culprit is an external connection, but it is physically possible to cause a leak, and even a disconnection, where the catheter connects to the button. The best solution to this is to make sure these forces don’t occur, but if warranted you can take extra steps to secure this connection:
- you can place a suture around the joint,
- you can slide a silicone sleeve around it, or,
- you can bond the catheter to the button or we can do this for you (but this has the disadvantage that the surgery must be done in reverse, potentially exposing the catheter tip to contamination as it is tunneled through the body).
For more information, see our blog post, What Does it Take to Disconnect a Rodent Catheter?
Video: Rat VAB™ Tether Connection
IV Self-Administration Equipment for Mice

Chambers
As with rats, the Med Associates mouse chamber, part no MED-307A-CT-B1, is ubiquitous. A responsive spring-based counter-balance such as SMCLA is a must to remove slack from the tether (Med Associates part no. PHM-124MA).
Tethers
Mice are one-tenth the weight of rats and so component design is critical. Only use 25ga swivels—mice cannot turn 22ga rat swivels without significant effort. Use the lightest possible springs; spring tethers designed for rats will restrain a mouse. If you find that the mouse is able to bite or climb up its spring tether, optimize the tether length and your counter-balance and that will usually solve the problem. There have been several options for catheter exteriorization and animal connection:
 |
Open lumen skin button (Instech 250P or LW62BS; distributed by Med Associates as PHM-124C). This product, which we designed about 40 years ago, was frequently used for mouse IVSA when there weren’t any other choices, but today it is a suboptimal option. Its biocompatibility is poor, the catheter connection is awkward and it is an open system. Open systems lead to subclinical and clinical infections, and short patency – impacting scientific outcomes and animal welfare. If you are still using these it’s time to upgrade. |
 |
Back-mounted pedestal (CamCaths MIVSA); a small version of the IVSA28 for rats, this device also is an open system and connection is awkward, though better than the LW62BS. Availability may be limited since CamCaths ceased operations. |
 |
Harness. We designed a miniature version of the Vascular Access Harness™ (Instech VAH62AB; Med Associates PHM-VAH62AB-P) with a small port for clean and simple connection, but the fact is that mice are not small rats. Mice do not tolerate harnesses well; their behavior changes noticeably and they are better than rats at escaping from them. |
 |
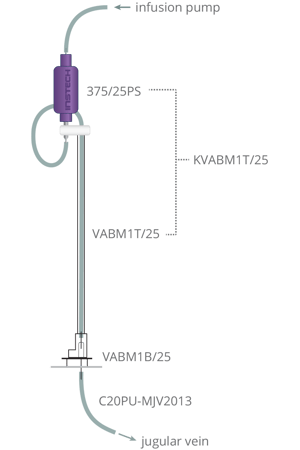
Vascular Access Button™ (Instech VABM1B/25; distributed by Med Associates as PHM-VABM1B/25). As with the VAB™ for rats this is an ideal catheter-exteriorization device for mouse IVSA. The subcutaneous catheter and closed system lead to extended patency. You can connect or disconnect the tether in seconds, and you can group house with the protective caps. Charles River reported 5x longer patency in mice compared to externalized catheters and Slosky et al at Duke reported a patency half-life of over 100 days and "unprecedented success in mouse self-administration approaches." |

Mouse Catheters

As with rats you should use sterile, polyurethane catheters with rounded-tips. Proper tip position, guided by a collar marking insertion length, is critical. For a typical 25g mouse, 2Fr (0.66mm OD) fits well in the jugular vein; a collar positioned at an insertion length of 13mm is common but may vary based on your surgical techniques, weight and strain. Our model C20PU-MJV2013 has these features as well as an inner diameter that is an ideal fit on the 25ga connector on the mouse Vascular Access Button™.
Beyond the Equipment: Surgery and Maintenance
None of these improvements in equipment will do you any good if you do not have a skilled surgeon on staff and the right space and equipment for him or her to operate. The basics include:
- A clean operating area, away from the traffic of your lab. The operating area is ideally separated from the preparation and recovery of the animals.
- A surgical microscope, and a good illumination system is a must for rodent surgery.
- Tools designed specifically for microsurgery, such as vessel dilator and the use of jeweler's forceps for tissue handling.
- Strict aseptic technique including pharmaceutical-grade flush and lock solutions, animal preparation, sterile drape material, such as Press'n Seal.
- Appropriate recovery. Heat implementation should be provided during recovery. It should also be provided at anesthesia induction and throughout the surgical procedure. Recover for at least five days. Some report significant improvements in weight gain when animals are group housed in recovery. Addition of palatable supplements may also support gain weight post-operatively.
Training. You’ll minimize trauma and time under anesthesia while optimizing tip placement and exteriorization with proper training and practice. If you do not have a skilled mentor, training opportunities can be hard to find but they do exist! Instech offers live online catheterization training for both mice and rats using a variety of techniques.
 If you are not able to set up for surgery - perhaps you just need to perform one or two IVSA studies as opposed to ongoing research - consider purchasing your rats or mice with the catheter and VAB™ implanted as a surgical service. Animals will arrive recovered from surgery and ready for use after they’ve been through your facility’s required acclimation period.
If you are not able to set up for surgery - perhaps you just need to perform one or two IVSA studies as opposed to ongoing research - consider purchasing your rats or mice with the catheter and VAB™ implanted as a surgical service. Animals will arrive recovered from surgery and ready for use after they’ve been through your facility’s required acclimation period.
Maintenance protocol. Avoid the temptation to flush the catheter just to maintain patency. This may be helpful with an exteriorized catheter, where evaporation will pull blood into the tip (be aware of laminar flow), but with the VAB™ we have consistently heard that less is more. Several studies run by Charles River support this, as they have demonstrated in mice and rats that catheters can be maintained long term by flushing low concentration heparin-saline (5-20 USP units/mL) no more than once weekly when animals are not on study. Always use positive pressure technique when accessing catheters.
Conclusion
Intravenous self-administration studies require the longest patency of any type of catheterized rodent study. Furthermore, these are behavioral studies, so the animals must be as healthy and as free of restraint as possible to give you the best data. These are not the kind of studies where you want to cut corners with homemade non-sterile catheters or a makeshift surgical suite.
Using a Vascular Access Button™ with a sterile, round-tip polyurethane catheter is a great start for both rat and mouse IVSA. The combination will give you the longest documented patency of any rodent device, the ability to group house, and quick, clean connection and disconnection. Once you have set up a reliable connection to the catheter, you will need to optimize your tether lengths and have a smoothly rotating swivel and sensitive counter-balance to minimize tether problems and its impact on behavior. Finally, just as important as the equipment, you will need aseptic access and maintenance with pharmaceutical-grade flush and lock solutions to achieve patency and successful study outcomes.




