A syringe pump is the standard device for delivering intravenous doses to rats and mice given its ability to dispense small volumes precisely. Most labs use workhorse laboratory syringe pumps, such as those made by Harvard Apparatus; they are simple to use, they’re built like a tank, they require very little maintenance (there is nothing to calibrate), and most labs have them lying around from a previous experiment. Others, notably the Contract Research Organizations (CROs) that are in the business of GLP infusion toxicology studies, prefer clinical syringe pumps that have a range of additional safety features such as syringe size detection and an occlusion alarm. However, as hospital nurses and lab animal technicians can attest, these alarms can be a huge pain in the neck. False alarms can be frequent, requiring needless intervention and documentation and an interruption in drug delivery. Why bother?
 |
 |
 |
| An infusion set leak caused by a pump without an occlusion alarm | Baxter AS50 | OrchesTA™ model 100 |
The answer is, if knowing that the correct dose was delivered into your animal’s vessel is important to you, a good occlusion alarm is essential.
Some researchers that use laboratory syringe pumps report that they never have a problem with occlusions. Of course not; the motor on the smallest Harvard Pump will keep turning until it generates 16kg of linear force. The weakest point of their infusion line will give way long before this and the test article will drop undetected into the cage bedding.
Unfortunately, many of the clinical pumps in use in CROs are older models such as the Baxter AS50, which sense occlusions by monitoring motor current. This is an indirect measurement: the motor will begin to struggle only after enough pressure builds up in the infusion line to start bending the plastic syringe plungers and plastic parts in the pump. This causes a delay in the alarm (in fact, the AS50 was subject to an FDA recall for this reason back in 2005 before it was discontinued), but it’s also the cause of false alarms: in order to detect occlusions in an acceptable time the sensitivity has to be such that normal friction in the pump can trip the alarm as well. This typically gets worse as the pumps wear with age.
A better approach, taken by modern clinical pumps, is to put a force transducer touching the syringe plunger, a more direct measurement of the pressure in the line. With less noise in the signal, the sensitivity can be set to an appropriate level that real blockages in flow are sensed quickly but false alarms are minimized.
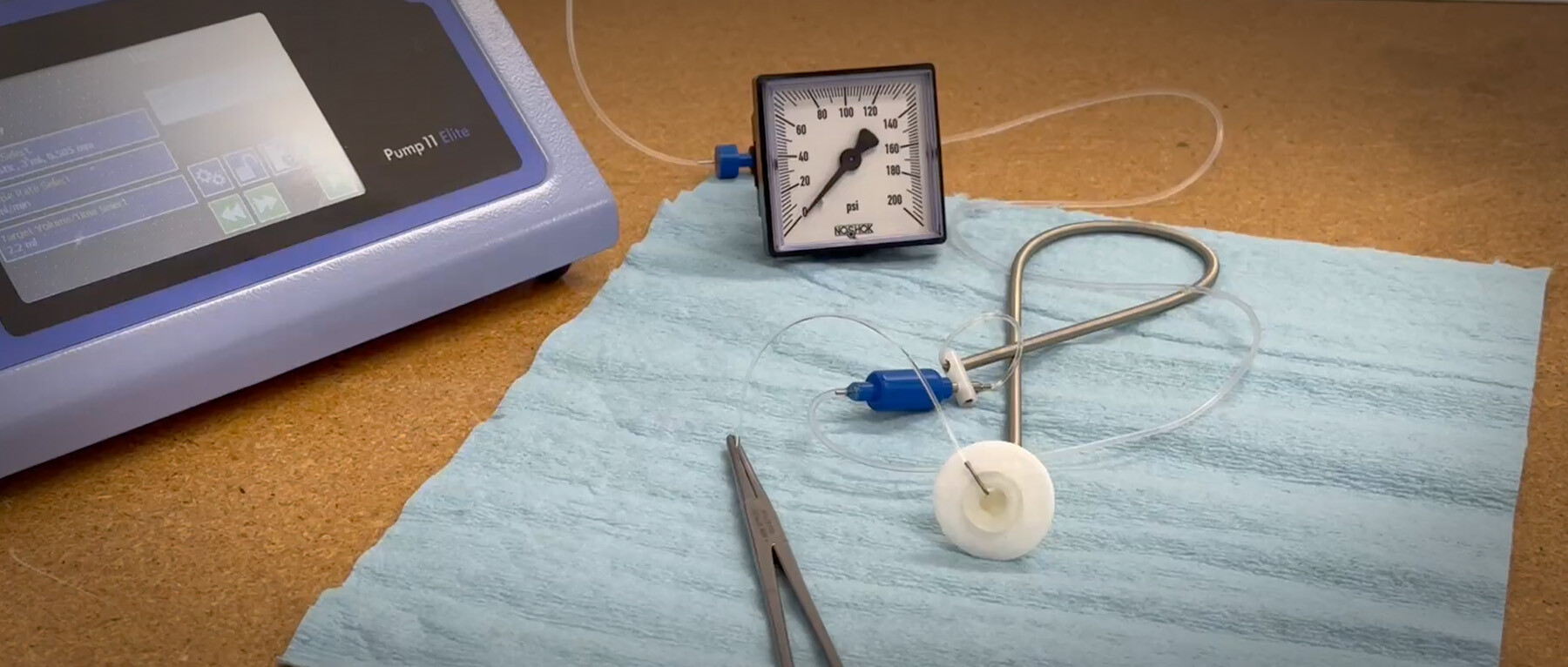
We conducted a study of how different pumps responded to a simulated blocked rat catheter at the end of a typical rat infusion tether part way through the study. The Harvard Pump never stopped, causing a leak and an undetected 75% dose error. The Baxter AS50 alarmed after 2 minutes and had a 20% dose error. The OrchesTA™ model 100 pump, a modern design, alarmed after 18 seconds, automatically backed off until the pressure was back to normal, and had a 1.5% dose error.
Patency issues or kinks in rodent catheters are a virtual certainty over a large multi-day infusion study. The differences between pump performance when there is an occlusion are dramatic. What can you do about it? If you work in a CRO, use modern clinical infusion pumps and test how the pumps (and your technicians) respond when there is an occlusion, not just how accurate they are in ideal conditions. If you are choosing a CRO for an infusion study and dose accuracy is important to you, ask what kind of pumps are being used in your study and make sure those pumps detect occlusions using a force transducer.
For more information, download our study on the impact of occlusions on dose accuracy.
DISCLAIMER: Instech sells both Harvard Apparatus and OrchesTA syringe pumps. The Baxter AS50 is discontinued.
UPDATE: See our November 2025 post which expands on this topic with new data: Syringe Pump Performance When There is an Occlusion.