- Applications
- Oral Gavage
- IV Drug Infusion
- Mouse Intermittent
- Mouse Continuous Infusion
- Mouse Continuous Infusion Plus Blood Sampling
- Mouse Two Channel Infusion
- Rat Intermittent
- Rat Continuous Infusion
- Rat Intermittent Infusion and Blood Sampling
- Rat Continuous Infusion Plus Blood Sampling
- Rat IV Bolus
- Rat Two-Channel Infusion
- Rat Infusion and Whole Body Plethysmography
- Rat GLP Infusion Toxicology Studies
- Large Animal Continuous Ambulatory Infusion
- IV Self-Administration
- Blood Sampling
- Glucose Clamp
- Bile Sampling
- Products
- Feeding Tubes
- Catheter Flush and Lock Solutions
- Catheters
- Buttons & Tethers
- Swivels & Mounts
- Tubing, PinPorts™ & Connectors
- Pumps
- Automated Blood Sampler
- Rodent Dose Puller
- Education
- Resources
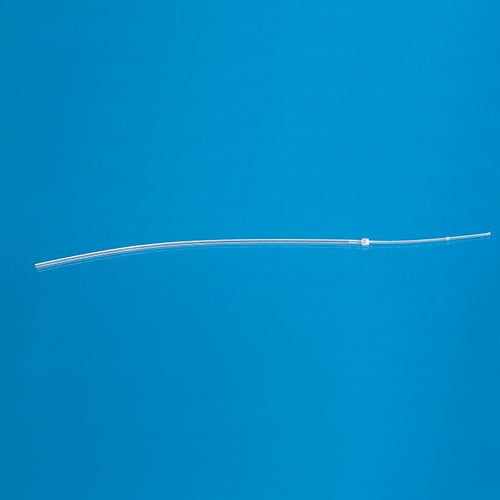
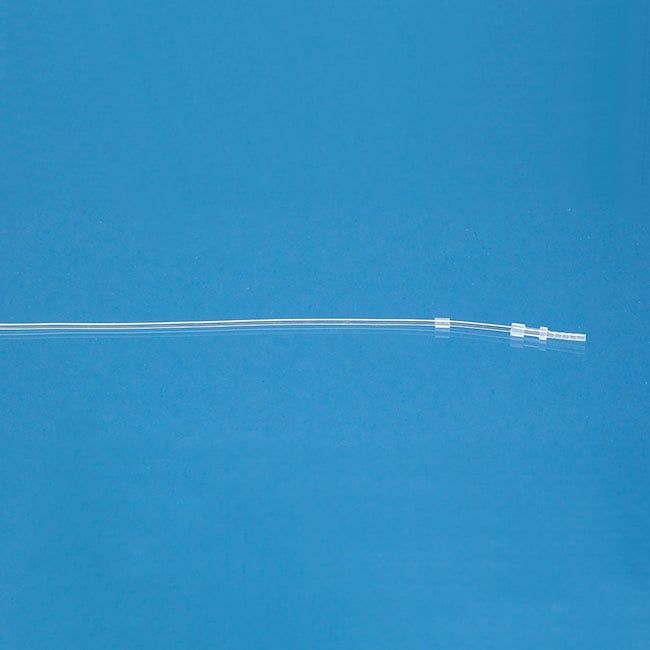
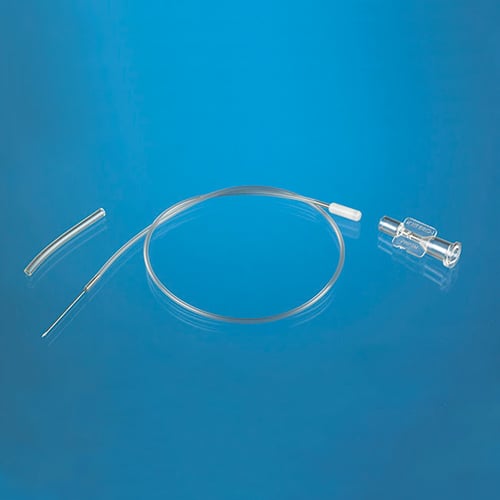
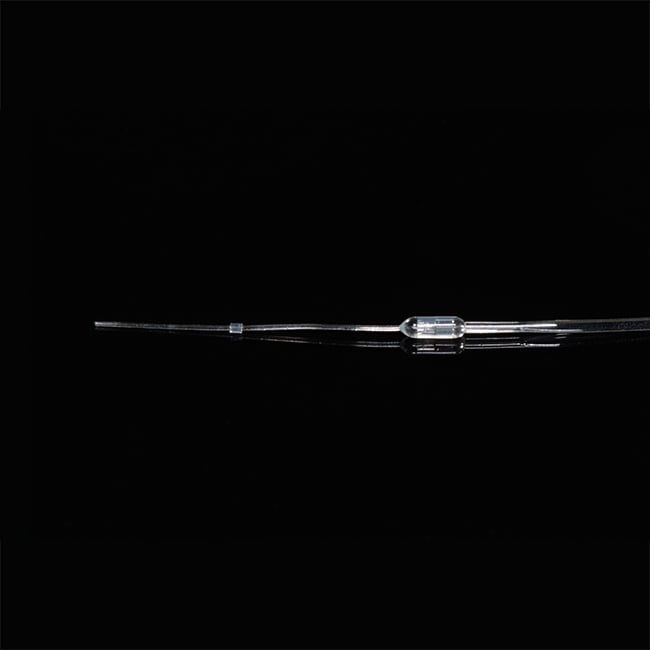
Catheters for Mice and Rats
Three principles drive the designs of our mouse and rat catheters: (1) that each model is designed specifically for the species and vessel; (2) that the catheter has an ideal fit with the external device to which it will be attached; and (3) that customization is simple enough that every surgeon can have a design to match his or her technique.
All our catheters are made of medical grade tubing, assembled and packed in a cleanroom and sterilized by ethylene-oxide gas. They are available in the following sizes (IDxOD):
3Fr – 0.64 x 1.0mm
2Fr – 0.43 x 0.69mm
1Fr – 0.18 x 0.36mm
0.8Fr – 0.13 x 0.25mm
Always use sterile lock and flush solutions, otherwise you have defeated the purpose of having a sterile catheter.
Filter Products
Frequently Asked Questions
What size catheters do you offer?
We offer catheters in the following sizes (inner diameter x outer diameter):
-
3Fr (0.64 x 1.0mm)
-
2Fr (0.43 x 0.69mm)
-
1Fr (0.18 x 0.36mm)
-
0.8Fr (0.13 x 0.25mm)
We can help determine the appropriate catheter for you based on your application, vessel, species, and area of research.
Are your catheters sterile?
Yes. All our catheters are made of medical grade tubing, assembled and packed in a cleanroom and sterilized by ethylene-oxide gas.
Can you provide the dead volumes for the catheters?
Specification sheets are available for each of our catheters within the “Ordering Information” table on Product and Application pages which include dead volumes.

Do you customize catheters for specific studies?
Yes. One of our catheter design principles is that customization is simple enough that every surgeon can have a design to match their technique. Contact Us for more information on how to request a custom catheter for your study.
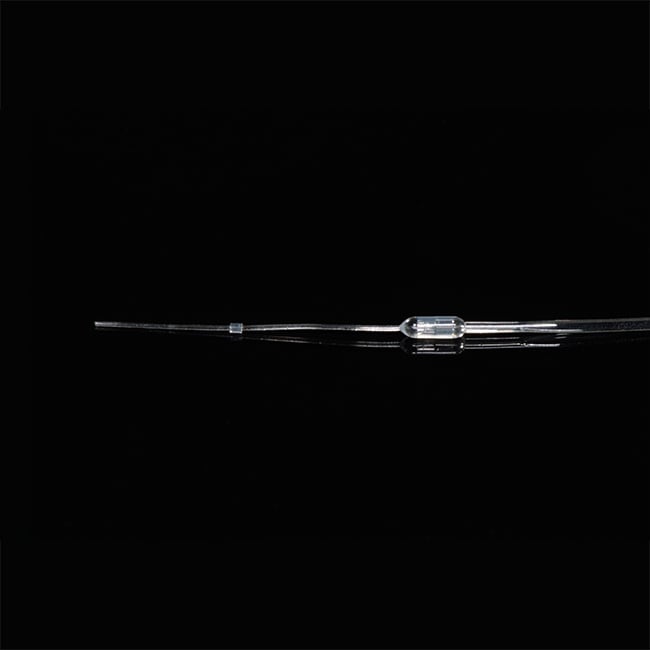
What shape are the catheter tips?
Instech catheter tips are rounded (unless otherwise specified) making them easier to insert, cause less trauma on the vessel wall, and help improve patency.
Does catheter tip shape make a difference?
Yes, catheter tip shape does matter. Rounded tips are easier to insert, cause less vessel wall trauma, and this study from Charles River shows they do lead to better patency.
Do you have any best handling practices for vascular catheters in rodents?
As with any catheterization procedure, particularly survival procedures, it’s important to follow proper aseptic technique to maintain sterility. Our Online Rodent Catheterization Training courses cover this in detail.
How often should I maintain my catheter?
When using a closed system such as with the VAB™, the recommended maintenance (using a sterile, pharmaceutical-grade flush and/or lock solution) schedule is seven days. If the catheter is exteriorized, or if using an open system, the catheter should be maintained every three to four days.
Is there anything that can be done to improve the patency of the catheter in studies?
There are five significant factors that influence patency: catheter tip placement, using a closed system, catheter material and tip profile, sterile components, and using the positive pressure technique. This blog covers each of these principles in detail.
Does the location of the catheter tip make a difference?
Implanted catheter tip location most certainly makes a difference—it’s critical! Actual tip location depends on a few factors. Our education program dives more deeply into the topic, but for a great overview read “Catheter Tip Location and its Impact on Patency.”